We Provide
Paracelsis is a preclinical (nonclinical) contract research organisation and toxicology consultancy serving the pharmaceutical industry
We provide lab-based,
in-house studies
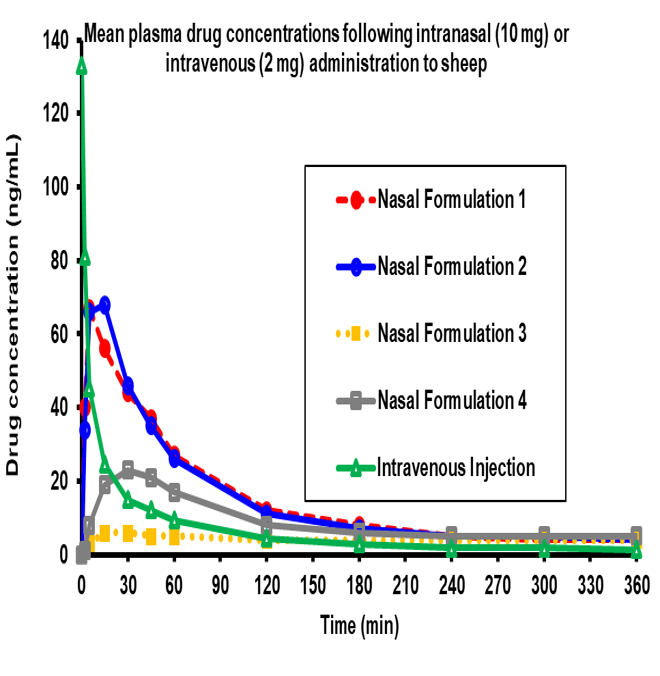
Our knowledge, expertise and knowhow in preclinical research and development allows us to offer a range of bespoke laboratory studies that can be integrated with your pharmaceutical product develpment plan. Many of these studies are designed to provide its customers with robust proof of concept data to support early decision milestones.
If you are interested in our laboratory-based studies and would like to find out more about how we can help you, please get in touch with one of our team today.
We can also help you to outsource studies
To support the full breadth of preclinical research and development of pharmaceutical products, Paracelsis can help its customers to outsource studies for example studies designed to meet Good Laboratory Practice (GLP) guidelines. Its pharmaceutical outsourcing services include: CRO selection, protocol generation, study monitoring and reporting.
Whatever your preclinical research requirements, please get in touch with one of our team today.

Why choose paracelsis for
your pharmaceutical research?
Bespoke Service
Our customers each have different R&D needs, which is why our services are tailored to your exact requirements.
One-stop Solution
We can guide and provide you with the necessary support from initial concept through to market approval of your product.
Read about our
toxicology consultancy services
Paracelsis operates a toxicology consultancy service primarily serving the Pharmaceutical Industry. However, our services are widely applicable across other Industries such as those involving Chemicals, Cosmetics, Food, Medical devices and Veterinary products.
Our services include toxicological assessment of active pharmaceutical ingredients (APIs) and/or their metabolites as well as excipients, impurities, degradants and extractables/leachables.
Added-value
Our range of consultancy services complement our CRO activities which provides added-value to our customers and fits within our ‘one-stop’ philosophy
Expertise when you need it
Our team are experts and many toxicological concerns can be answered following a thorough review of the literature thereby saving time and money
Avoid Unnecessary And Costly Testing
In circumstances where published literature are inadequate, we can undertake in-silico (e.g. (Q)SAR) assessment thereby helping to avoid further, unnecessary, laboratory testing